Results for 'diagnosis'

Magnitude of Clinical Benefit of Cancer Drugs Approved by the US Food and Drug Administration
antineoplastic agents, immunologic adjuvants, pharmaceutical adjuvants, phase 3 clinical trials, drug approval, drug labeling, medical oncology, united states food and drug administration, diagnosis, palliative care, surrogate endpoints, weight measureme
Regulatory agencies assess drug safety and efficacy, but thresholds may differ from those accepted by clinicians . Only 43.8% of RCTs for FDA-approved drugs meet the ESMO-MCBS threshold for meaningful benefit, reflecting potential softening of FDA standards. Encouraging trends include an increas…
Dec 13th • 7 mins read

Transforming oncology: Five frontiers driving progress in cancer care
From biomarker-driven breakthroughs to AI-powered early detection and a renewed commitment to equity and patient centricity, the past 12 months have seen major strides across cancer research, treatment, and communication. At Inizio, we’ve had a front-row seat to this transformation, supporting…
May 16th • 5 mins read
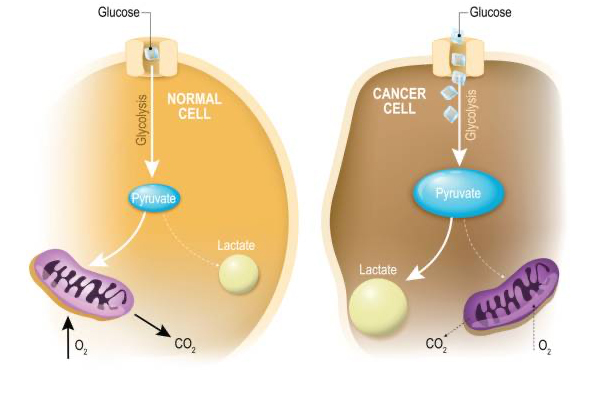
“Oncometabolism: The switchboard of cancer: An editorial”
Otto Warburg, biochemist, FDA, FDG-PET, MRS, Oncometabolism
Otto Warburg's discovery in the 1920s highlighted a unique bioenergetic phenotype in tumor cells, emphasizing their reliance on lactic acid fermentation, which spurred the field of oncometabolism. Oncometabolism has led to advancements like FDG-PET imaging for cancer diagnosis and staging,…
Feb 1st • 1 min read

Advances and Challenges in Pediatric and Childhood Cancers
CNS, central nervous system, ES, Ewing sarcoma, MB, medulloblastoma, PTSD, post traumatic stress disorder, RB, retinoblastoma
Pediatric cancers, while uncommon compared to adult cancers, include leukemia, CNS tumors, lymphoma, Wilms' tumor, and retinoblastoma. Challenges in pediatric cancer treatment encompass late diagnosis, varied immune responses, preserving fertility, minimizing late effects, and considering lo…
Jun 27th • 2 mins read
Liquid biopsy in oncology: a consensus statement of the Spanish Society of Pathology and the Spanish Society of Medical Oncology
Liquid biopsy, ctDNA, Oncology, Genomic profling, Precision medicine
The proportion of cancer patients with tumors having potentially targetable genomic alterations is increasing. Diagnosing these alterations can lead to tailored treatments and provide additional predictive information on immunotherapy efficacy. In many cases, initial tissue biopsies are ina…
Sep 26th • 17 mins read

Pediatric Oncology Clinical Trials and Collaborative Research in Africa: Current Landscape and Future Perspectives
pediatric oncology, clinical trials, research, Africa, collaborative research
Current childhood cancer survival rates in African countries range from 8.1% to 30.3%. The review focuses on pediatric oncology trials in Africa, highlighting challenges and suggesting research collaboration opportunities. Data were collected from the SIOP global mapping survey, ClinicalT…
Aug 7th • 10 mins read
Pivotal Considerations for Optimal Deployment of Healthy Volunteers in Oncology Drug Development
drug development, healthy, volunteers, oncology clinical trial
The content discusses the challenges and opportunities in conducting oncology clinical trials, particularly focusing on trials involving normal healthy volunteers (NHVs) and patients with cancer. There are over 5,000 ongoing oncology trials in the U.S., with low enrollment rates among adult c…
Oct 31st • 20 mins read

Liquid biopsy in oncology: a consensus statement of the Spanish Society of Pathology and the Spanish Society of Medical Oncology
Liquid biopsy, ctDNA, Oncology, Genomic profling, Precision medicine
The proportion of cancer patients with tumours that have targetable genomic alterations is increasing, which is crucial for tailored treatment and predicting immunotherapy efficacy. Initial tissue biopsies are often inadequate for precision oncology, and obtaining new tumour tissues at pr…
Sep 26th • 16 mins read

Level of evidence used in recommendations by the National Comprehensive Cancer Network (NCCN) guidelines beyond Food and Drug Administration approvals
oncology, guidelines, off-label drug use
The analysis reviewed 113 NCCN recommendations, focusing on 44 off-label uses of drugs. 14 of these off-label recommendations were later FDA-approved or backed by RCT data. 13 recommendations were minor extrapolations from the FDA label or actually on-label. Of the remaining 17 extrapolations…
Aug 2nd • 8 mins read
Representation of Patients With Cardiovascular Disease in Pivotal Cancer Clinical Trials
cancer, cardiovascular disease, clinical trials, underrepresentation, United States Food and Drug Administration, FDA, CVD
Heart failure (28%) was the most common exclusion criterion in clinical trials for FDA-approved anticancer therapies, followed by prior coronary disease and arrhythmias (27% and 23%, respectively). Over one-third of trials excluded patients with underlying cardiovascular disease (CVD), including mor…
Mar 18th • 3 mins read
